(Continued from Ch2/part1)
Dam you straight to heck
This issue makes the case that it is both necessary and possible to reduce our resource and energy use. In Chapters 3 and 4, we look at concrete ways that we can do so while improving our quality of life. However, before making that case, we have to more thoroughly establish why it is necessary to reduce our energy and resource use. That can’t be done without fully illustrating why better technology, regulation, and monitoring are inadequate solutions to the problem of mining waste. In other words, we will demonstrate in this section that the only viable solution to mining waste is to do less mining. This is a problem we cannot afford to ignore: according to the most recent (2000) estimates, there are 3,500 tailings dams in the world, and a full green energy transition will require thousands more.
____________________________________
Issue 2: Green energy & electric cars
Intro: Why does green energy produce so much toxic waste?
Chapter 1: Comparing wind, solar, nuclear & geothermal energy; mining waste
Chapter 2: The human and environmental costs of green energy mining
Chapter 3: Phasing out cars (even electric ones) to save the planet
Chapter 4: Your life would be way better if we phased out cars
Bonus 1: Planet-saving green energy technology we foolishly never developed
Bonus 2: How did Congo (the world’s leading cobalt producer) get the way it is?
Bonus 3: The Congo Wars (1996-2003) and its millions of victims
_____
Issue 2 is available in written and podcast format
__________
Above, we saw examples of catastrophes resulting from tailings impoundment failures: people killed, homes wiped out, and irreparable ecological damage. But what actually is a tailings impoundment? How are tailings stored away?
Tailings impoundments are massive earthen rings that encircle mining waste. Modern tailings impoundments are among the largest human-made structures on earth. The largest tailings impoundments are more than a mile thick and more than 40 feet tall, snaking through the landscape in a giant circle more than 10 miles around. Such massive structures require more than half a cubic kilometer of construction material, or enough to cover an area the size of Chicago with more than a meter of construction material. As miners clear away valueless rock to access mineral-rich rock, they dump it into the earthen ring. Waste from processing ore – corrosive chemicals that can dissolve rock – is also added. As more and more waste piles up, the impoundment is built higher and higher accordingly. Once the mine is exhausted, the top of the tailings impoundment is covered with earth. In this way, toxic tailings are encased in earth and isolated from the outside world. This description may make tailings impoundments sound simple, but in reality, they are complex technological wonders. From accurate subsurface analysis to choosing the construction material most appropriate for conditions, from engineering design to the actual construction, an extraordinary amount of planning, knowledge, and work goes into tailings impoundments. Tailings dams generally cost tens to hundreds of millions of dollars to build, and modern mines produce hundreds of millions of metric tons of tailings waste.
There are better and worse designs for tailings dams, but we need to be realistic about what can actually be accomplished when faced with such a difficult technological problem. Tailings impoundments must keep corrosive chemicals and individual toxins locked away, forever. How can we possibly design a structure that corrosive chemicals will never seep out of? How can we design something so perfectly solid that toxins like lead and uranium – at the level of an atom – can never find a hole tiny enough to slip out of?
A common belief is that better technology and better regulation can clean up any type of pollution. This cannot be squared with real-world experience. If better technology led to improved tailings dams, then brand new tailings dams with the latest technology wouldn’t collapse; they have. Similarly, better monitoring should be able to identify problems and prevent failures. In reality, tailings dams have failed while they were still being filled, when mining personnel were everywhere. It’s also not true that the free market can fix this problem: prices of minerals are expected to rise due to the demand of the green energy transition, and higher mineral prices should mean more money available to build better tailings impoundments. Yet the decade 2000-2010 saw both the highest mineral prices in a century and a substantial increase in high-severity tailings dam failures. Higher mineral prices, in other words, do not lead to safer tailings impoundment. Finally, it’s also not true that better regulation can fix the problem: tailings dams have failed in countries like Canada, Spain, Finland, and Sweden – hardly countries with unusually lax environmental regulation or enforcement. We can’t innovate or regulate our way out of mining waste.
This is especially true because tailings dams cannot be significantly improved after they are filled. There is no option of doing the best we can today and coming back with more resources and better technology years later: just as a swimming pool must be drained to make repairs, construction crews cannot SCUBA dive into toxic tailings and reinforce miles and miles of a dam that is forty feet tall.
This point is driven home with the so-called upstream tailings dam construction (see the graphic below). It is by far the cheapest design but obviously less safe: In an upstream construction, the tailings themselves are used to structurally support the dam, even though tailings are not construction material and are not optimized for structural stability (and some tailings are liquid(!)).
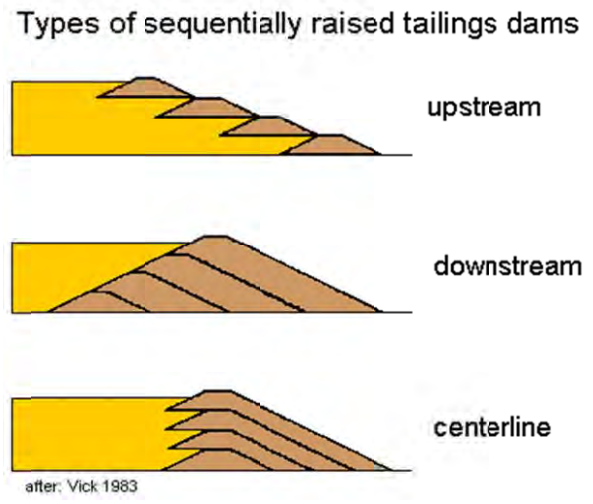
Yet downstream construction – while safer – is not safe. Though a rough calculation (because it includes tailings dams of different ages), an estimated 5% of upstream tailings dams have failed, and 2-3% of centerline and downstream tailings dams have failed.
These numbers are not reassuring. If 2% of all tailings dams fail every 40 years, all will have failed within 2000 years, leading to thousands of catastrophes like Germano. Considering the massive size of tailings impoundments – hundreds of millions of tons of toxic tailings, 10 miles around, forty feet high – and the mind-boggling timescales, it becomes clear why there is no amount of innovation that can solve this problem.
____________________________________
Featured Excerpts
Apocalypse Nord (Iss1/Ch4)
These policies led to the Great Depression…what could go wrong? (Iss1/Ch3)
Independence* Day (Iss1/Ch2)
Are we measuring global poverty or intentionally underestimating it? (Iss1/Ch1)
__________
__________
Weather forecast for the next 200,000 years
Tailings dams don’t suddenly fail; there is usually a precipitating event – something that overwhelms the dam. Though the data are incomplete, the most frequent precipitating event is extreme weather, and the second most frequent is an earthquake (see the Center for Science and Public Participation’s (CSP2) database of failures and this scholarly study of causes). If so much rain falls that it overwhelms the impoundment’s drainage system, the weight of the water can overwhelm the strength of the dam, especially if the water has seeped into the earthen dam and softened it. And, an earthquake simply collapses a portion of the dam in the way earthquakes destroy any human-made structure, as has occurred repeatedly in copper mine tailings dams in Chile.
Thus, to design a safe tailings dam, we must ensure that – forever – there will never be an earthquake large enough, or extreme weather event forceful enough, to breach the dam. This is fantasy. For earthquakes,
Seismologists know that there are many active faults that have not been mapped or have been mapped inaccurately, that some faults believed to be inactive may actually be active, and that there are many inactive faults that may become active again.
With the very real possibility that a tailings dam is built directly on top of an undiscovered fault line – a virtual certainty given that thousands of tailings dams already exist and thousands more needed for a full green energy transition, plus the geologic timescale tailings dams must remain intact – CSP2 recommends that the only safe option is to design all tailings dams to withstand an earthquake “that ruptures the ground surface on which the dam is built.”
Moreover, the authors of the report point out that the earthquake that caused the Fukushima power plant disaster in Japan was eight times more powerful than seismologists had estimated to be the most powerful earthquake that fault could ever produce. We simply don’t understand seismology well enough to make the necessary estimates to ensure each tailings dam can withstand an earthquake; we certainly don’t understand seismology well enough to accurately predict earthquakes for hundreds of thousands of years and beyond.
Likewise, the idea that we can confidently predict the strongest weather event a tailings dam will ever experience, hundreds of thousands of years into the future and beyond, is similarly fanciful. 10,000 years ago was the most recent Ice Age. We can’t possibly predict what the climate will be like in a specific area in a few thousand years; we obviously can’t predict individual weather events. How can engineers design for problems we can’t foresee?
The issue of changing climates and extreme weather is made more acute by climate change. The climate is already changing in unpredictable ways, and extreme weather events have already become more probable. In other words, the technological and engineering challenge of tailings impoundment is getting even harder to solve.
Would you like your poison solid or liquid?
CSP2 recommends (more detail here and here (p10)) that all tailings be solid. That’s because when a tailings dam fails, liquid tailings spill out over an enormous area, causing catastrophic, unremediable damage like at Germano. By contrast, solid tailings will mostly stay in place. In fact, CSP2 recommends that the tailings be structurally stable without the dam; in other words, the dam should simply be there to prevent the tailings from coming into contact with the outside world and protect it from wind and water erosion, but the dam isn’t actually necessary to hold the tailings in place. If this can be achieved, the advantage is obvious: failure of a solid tailings dam causes no harm as long as the breach is quickly repaired.
_________________________________
Most recent issues
Issue 1 | Climate change and drinking water
Issue 2 | Green energy and electric cars
______________________
Unfortunately – aside from being dramatically more expensive – there is a serious drawback of solid tailings, called acid mine drainage. Liquid tailings can be treated so that toxic heavy metals cannot escape: the technical term is “chemical stability.” We normally think of metals as a solid, but metals actually dissolve in water that is acidic. This is a problem because when heavy metals are dissolved in water, they can leach out of a tailings impoundment and into groundwater.
However, if a basic reagent is added, it lowers the acidity (raises the pH) of the tailings and the heavy metals “precipitate” – turn solid. In a basic environment (a pH of about 9), the metals will remain solid and will therefore be unable to leach out of the tailings dam. However, this process is only possible in liquified tailings; there’s simply no way to get a basic reagent mixed into solid tailings. This is the drawback of solid tailings: since the tailings cannot be made basic, toxic heavy metals are certain to slowly leach out of the dam and make their way into groundwater. In other words, the groundwater around a tailings dam with solid tailings will always be poisoned, and anyone who ever drinks from this groundwater will have to treat it to remove the heavy metals, forever.
The unappealing choice is thus between a catastrophic failure and a slow failure. Liquid tailings don’t leach heavy metals, but they do risk catastrophic failure, sending toxic waste over enormous expanses of land all at once. Solid tailings do not fail catastrophically, but they are certain to continuously, albeit slowly, release toxins into the groundwater. As we saw above, we must expect that all tailings dams will eventually fail. For this reason, CSP2 argues that the lesser of two evils is solid tailings – particularly since a single modern mine produces hundreds of millions of tons of mining waste, or so much tailings that the environmental damage of a liquid tailings dam failure is literally unremediable.
Nonetheless, we need to take seriously the fact that solid tailings will impose serious harm on future generations. Tailings dams need to be monitored for problems in order to prevent failure. If a solid tailings impoundment is breached, it must be quickly repaired, or wind and water erosion will spread toxic tailings far and wide. But how do we know that people 1000 years in the future will still know where all the solid tailings impoundments are and have the capacity to monitor, maintain, and repair them? If you think this is a trivial problem, think back to the ancient history thought exercise in this issue’s introduction.
Similarly, will people 1000 years from now know the groundwater around all these thousands of tailings dams is not safe to drink and how to treat it? In 2000 years? 50,000 years? 100,000 years? As discussed in the introduction, 7500 years ago, the very first cities were being settled, and 9000 years ago, pottery was being invented. We cannot possibly know that humanity will remember where all of these thousands of tailings impoundments are located, let alone have the ability to monitor, maintain, and repair them.
Another major issue is that groundwater doesn’t always stay as groundwater. Groundwater is water that has seeped into the soil, up to hundreds of feet deep and across endless miles of Earth’s surface. Toxins dissolved in groundwater thus cannot be removed with any technology because doing so would require somehow pulling all the water out of the ground over countless square miles, hundreds of feet deep into the earth. Fortunately, unless accessed by humans as drinking water, toxic groundwater is not dangerous. But groundwater can become surface water (like a river or lake) if it is forced upwards. The source of all rivers is either melting snow or groundwater that has been forced to the surface. CSP2 cautions that a mine should never be approved if the groundwater in the area becomes surface water.
But the timescales involved are enormous. Climatic or geological changes could alter the behavior of groundwater. Groundwater confined underground today may eventually be forced up to the surface by changes to climate or geology that we cannot predict. We can’t possibly be certain that leachate from a solid tailings impoundment will stay deep underground as groundwater, forever.
The reason we are facing a climate crisis is that people in positions of power thought that people living many years in the future – us – don’t matter. If we accept the perpetual poisoning of landscapes via acid mine drainage, we’re engaging in the very same thinking that got us into this mess in the first place. Whether liquid or solid, tailings are a clear danger to future generations, and we should not address the climate crisis with the same disregard for future generations that caused the climate crisis in the first place. Moreover, we certainly cannot claim to be “saving the planet” from climate change if we’re poisoning the very earth we are claiming to protect.
In situ mining
Clearly, there is no solution to mining waste: there is no technological solution that can confidently contain tailings for the necessary timescales, no regulatory solution that can ensure prudent monitoring and maintenance. But what if there were a different way to mine that did not generate so much waste?
In situ leach mining eliminates much – but not all – mining waste. In this section, we investigate uranium mining to establish the principles of in situ mining.
In the 1960s, in situ leach mining of uranium was attempted in the US and the Soviet Union; today, most uranium mining worldwide occurs via in situ methods, including in the US. In situ leach mining does not resemble traditional mining at all: there is no excavation whatsoever. Rather, small holes are drilled deep into the ground and chemicals are pumped in; these chemicals dissolve the uranium, and miners collect the uranium-rich solution through a different borehole, sucking the solution up like a straw. The solution is then taken off-site to extract the uranium from the solution and turn it into a usable form.
The least environmentally harmful process uses carbonate ions, the same component that makes the bubbles in sodas. When carbonate ions encounter uranium, a series of chemical reactions occur that eventually create U3O8, which is soluble in water.*
In situ mining is not possible for all uranium deposits. The rock must be porous enough to pump the chemical solution through it. In situ is also impossible if the uranium is surrounded by compounds that react with carbonate ions. Thus, much uranium ore can only be accessed via traditional mining.
Because there is no excavation, in situ leach mining generates substantially less waste overall and is substantially less harmful than traditional mining. In situ also eliminates the occupational hazards of mining, from breathing in toxic dust or gases, to the use of explosives and the danger of tunnel collapses. Nonetheless, in situ is not without significant harm. First, the chemicals used are not specific to uranium; they dissolve any heavy metal. In other words, the liquid drawn up contains not merely uranium, but a host of other toxic heavy metals, some of them also radioactive. Thus, once the uranium is processed, it leaves behind a toxic, radioactive soup of heavy metals. In other words, though in situ doesn’t generate tailings, it does generate a more concentrated waste that needs to be dealt with.
Second, the source of carbonate is often ammonium carbonate, which generates the pollutant NO3 in large quantities. Ammonium carbonate consists of two molecules of ammonium for every one molecule of carbonate. Thus, every carbonate generates two molecules of NO3. This huge amount of NO3 is not easily disposed of.
Finally, it is not possible to remove all the chemical solution forced into the rock. All unrecovered chemical-bearing solution will eventually become incorporated into groundwater, along with whatever it has dissolved. This – obviously – poisons the groundwater with heavy metals – some of them radioactive – and other toxins (such as NO3), rendering a once-clean source of water undrinkable.
Groundwater contamination remains a serious problem for in situ leach mining, despite decades of research into remediation. Ruiz et al state that “When feasible, [in situ leach mining] greatly reduces waste generated by the mining and milling processes, however, the ability to restore ground water to acceptable quality after [mining] ends is uncertain.”** The Black Hills Clean Water Alliance states more succinctly (though no less correctly), “Water at an in situ leach uranium mine has never been returned to its original condition.”
Sign up here to be notified when a new issue of Finite is published (4 emails/year)
In sum, in the limited cases where in situ methods can be used, it is substantially less harmful than conventional mining as it does not require excavation, thus generating substantially less waste. The major problem with in situ is that it is impossible to recover all the solution injected into the ground, so it contaminates groundwater permanently. Of course, traditional mining also poisons groundwater; the point is that there is no environmentally friendly form of mining. There are more and less harmful ways of mining, but there is no such thing as harmless mining.
Conclusion: the type of thinking that got us into trouble can’t be the thinking that gets us out
Chapter 1 started out as a duel between nuclear, wind, solar, and geothermal energy to see which was the best option to phase out fossil fuels. This exercise led to some surprising conclusions. Geothermal was surprisingly competitive. The large difference in resources required was impressive, with a single nuclear power plant able to produce as much electricity as 800 wind turbines or 8.5 million solar panels.
But the further we got, the clearer it became that debating the merits of different forms of green energy was missing more fundamental questions. When we rushed headlong into planning out our green energy transition, we failed to ask more fundamental questions about how our world should work. For example, we never considered the possibility that perhaps we don’t need all the energy we think we do. Indeed, fossil fuel corporations are corporations: as discussed in Iss1/Ch4, they are legally bound to pursue profits above all other considerations. More energy use means more profits, and given the massive political, economic, and social clout held by fossil fuel corporations, it would be shocking if we were not using more energy than we need. This matters greatly because, as we saw here in Chapter 1, mining waste imposes severe environmental impact. If we can reduce our energy needs, we can prevent literally billions of tons of mining waste from being created. In Chapters 3 and 4, we study one dramatic example of how we can massively reduce our energy use while improving our quality of life. But first, Chapter 2 examines the mining processes for some of the critical minerals of the green energy transition.
*An alternative chemical process has never been approved in the US due to even greater difficulty with groundwater remediation compared to the carbonate method.
*The most promising attempts at groundwater remediation involve using bacteria that naturally live in extreme environments with lots of toxic heavy metals. These bacteria have evolved ways of turning dissolved heavy metals into solid heavy metals; once solid, they are no longer harmful. In principle, if these bacteria are introduced into an aquifer that has been contaminated by in situ mining, they should be able to remediate the water. Wufuer et al point out that:
Among the uranium bioremediation techniques available, uranium bioreduction has been studied extensively, where U(VI) is reduced to U(IV) by a variety of bacteria. In situ demonstrations of uranium bioreduction have been conducted successfully at the US DOE Rifle site, Colorado [27], and at the US DOE Oak Ridge site, Tennessee [28,29]. However, the stability of reduced U(IV) depends on protecting it from re-oxidation by nitrate [30,31] and O2 [28].
Ruiz et al summarize non-bioremediation attempts (which have not proven successful) and overall agree that bioremediation may not be permanent:
Newsome et al. (2014), in an extensive review, summarized the results of field studies in which in-situ bioremediation of U was investigated at the Rifle, CO mill tailings site, and ground water contamination at sites in Oak Ridge, TN and Hanford, WA. The studies have found that while in-place immobilization of U can be achieved through microbial reduction, this effect may be temporary with subsequent release of U and other constituents after organic feed is discontinued or after re-oxidation.