When the Indian Point nuclear power plant was closed in 2021, it was cause for celebration among some environmentalists. The Natural Resources Defense Council tweeted, “It’s been an honor for NRDC to work with our tireless partners and advocates who have fought long and hard to close Indian Point.” Alec Baldwin opined, “Let’s raise a glass to everyone who was brave and worked hard” to shut the plant down, citing concerns over the disposal of spent nuclear fuel. Apparently, concern over nuclear waste was so strong that it dwarfed concerns over climate change: while nuclear power emits no greenhouse gas and thus does not contribute to climate change, Indian Point was replaced by three fossil fuel power plants. Clearly, some environmentalists consider nuclear energy to be worse than fossil fuels due to the radioactive waste of spent nuclear fuel. As we saw last issue, climate change has the potential to cause mass extinction and reduce the human population to half a billion people. What is so dangerous about nuclear waste that it exceeds the dangers of climate change?
____________________________________
Issue 2: Green energy & electric cars
Intro: Why does green energy produce so much toxic waste?
Chapter 1: Comparing wind, solar, nuclear & geothermal energy; mining waste
Chapter 2: The human and environmental costs of green energy mining
Chapter 3: Phasing out cars (even electric ones) to save the planet
Chapter 4: Your life would be way better if we phased out cars
Bonus 1: Planet-saving green energy technology we foolishly never developed
Bonus 2: How did Congo (the world’s leading cobalt producer) get the way it is?
Bonus 3: The Congo Wars (1996-2003) and its millions of victims
_____
Issue 2 is available in written and podcast format
__________
To understand what these environmentalists were so concerned about, it is necessary to understand how nuclear power works. Nuclear power plants use a special type of uranium – uranium-235 – as fuel. Once all the uranium-235 is used up, it leaves a mess of dozens of different elements, all of them radioactive. While nuclear power has the advantage of emitting no greenhouse gases, this must be weighed against the dangers of spent nuclear fuel.
But it turns out that radioactive waste is not a problem unique to nuclear power. Any path we choose to phase out fossil fuels must contend with a giant amount of radioactive waste. As we’ll see in Chapter 1, generating 1 gigawatt of electricity from nuclear power leaves behind 276 tons of nuclear waste, but generating 1 gigawatt of electricity from wind power leaves behind 28,000 tons of radioactive waste.
The atoms trying to kill you
Why would seemingly clean forms of energy, like wind and solar, generate so much waste? The biggest contributor is mining. To obtain the critical minerals needed for a green energy transition – lithium, cobalt, nickel, rare earths, copper, etc. – requires a great deal of mining, and thus generates a great deal of mining waste.
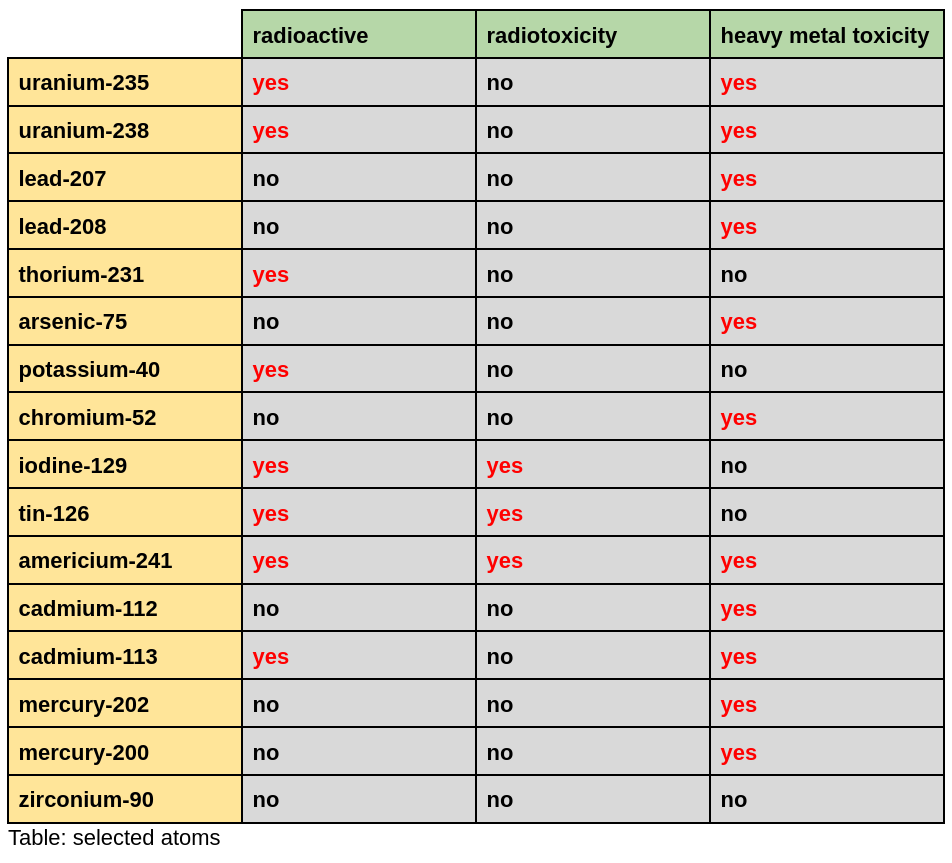
Surely nuclear waste is more dangerous than mining waste. Or is it? To make an accurate comparison, there are three terms we need to understand: radioactivity, radiotoxicity, and heavy metal toxicity. Let’s take these in turn.
Heavy metal toxicity is the easiest to understand. Heavy metals like arsenic, lead, cadmium, and chromium interfere with the normal function of cells – the building blocks of all life – and are thus toxic to every part of the body.
Radioactivity requires more scientific context to understand. As we learned in middle school science class, atoms consist of a tiny nucleus of protons and neutrons surrounded by a cloud of electrons. Radioactive atoms are dangerous because their nucleus is unstable. All radioactive atoms eventually undergo the natural process of radioactive decay, wherein the nucleus adjusts to a more stable configuration. This decay process releases nuclear radiation, which is harmful to living things in large enough doses. For example, when uranium-235 (92 protons + 143 neutrons = 235) undergoes radioactive decay in nature, it transforms into thorium-231 (90 protons + 141 neutrons = 231) and releases alpha radiation (an alpha particle is 2 protons + 2 neutrons).
While all radioactive atoms have an unstable nucleus, radioactive nuclei exist along a spectrum with a huge range of instability. Instability is measured in “half-life,” or the amount of time that would pass before half of a large amount of radioactive atoms had undergone their natural decay process. For example, lanthanum-138 has a half-life of 102 billion years, so if you had 10 pounds of pure radioactive lanthanum-138, it would take 102 billion years for five pounds to undergo the natural radioactive decay process. By contrast, in the upper atmosphere, fluorine-19 can turn into fluorine-20 when struck by cosmic radiation. Fluorine-20 is so unstable that it decays almost immediately: its half-life is just 11 seconds.
Crucially, an atom can be radioactive but not radiotoxic. For a radioactive atom to be radiotoxic, it must be radioactive in a way that is dangerous. For example, uranium-238 is radioactive but not radiotoxic: at 4.5 billion years, its half-life is so long that even if you ingested some, you would die of old age long before enough decayed to give you a dangerous dose of radiation. Americium-241 is radiotoxic because its half-life is only 433 years. Though 433 years may seem long, the half-life is sufficiently short that – if ingested – enough americium-241 atoms would decay over a human lifespan to give a dangerous dose of radiation. Americium-241 is a useful example because it is found in nearly every room of every home: americium-241 is an essential component of smoke detectors, illustrating that radioactive and radiotoxic atoms can be safely managed.
But half-life is not the only factor determining whether a radioactive atom is radiotoxic. Radioactive iodine-129 is dangerous even though it has a very long half-life of 15.7 million years because the human body stockpiles iodine in the thyroid. Concentrating all of the radiation in a small part of the body means iodine-129 is radiotoxic, despite its long half-life. Tin-126 also has an extremely long half-life of 230,000 years, but it decays into antimony-126. Because antimony-126 is highly radioactive, tin-126 is radiotoxic.
While atoms can be radioactive but not radiotoxic, some atoms are both radioactive and heavy metal toxic. For example, as discussed above, uranium is radioactive but not radiotoxic. However, uranium is toxic in even low doses as a heavy metal. By contrast, thorium-231 is radioactive but not toxic as a heavy metal. Lead-207 and lead-208 are toxic heavy metals but are not radioactive. One out of every 10,000 atoms of potassium is radioactive potassium-40, but potassium-40 is neither radiotoxic, due to its long half-life of 1.2 billion years, nor heavy metal toxic.
It may seem logical that radiotoxicity is more dangerous than heavy metal toxicity. This is not true. While there is a wide range of danger resulting from both radiotoxicity (all forms of antimony are more dangerous than americium-241) and heavy metal toxicity (all forms of mercury are more dangerous than iron), it is not true that exposure to radioactivity is always more dangerous than exposure to heavy metals.
With this background, we can start to compare the dangers of nuclear waste versus mining waste. There are three key points of difference.
#1 Much nuclear waste eventually becomes safe, but mining waste doesn’t
100% of nuclear waste is radioactive, and more than 98% of nuclear waste is radiotoxic. But as discussed above, all radioactive atoms undergo a natural process of radioactive decay, reaching a more stable configuration. In other words, all radioactive waste will eventually cease to be radioactive. For nuclear waste, within 300 years, 97% of all radioactive atoms have undergone their natural decay process and are no longer radioactive. Thus, close to 100% of the radiotoxic atoms in spent nuclear fuel are no longer radiotoxic within 300 years.
The end of radioactivity does not mean that nuclear waste is rendered harmless. For example, uranium-238 and thorium-221 decay several times before reaching a stable configuration, but unfortunately, that stable configuration is the toxic heavy metal lead (lead-207 and lead-208, respectively). On the other hand, some atoms in nuclear waste do become harmless after undergoing radioactive decay. For example, strontium-90 eventually decays to zirconium-90, which is not radioactive and is not a toxic heavy metal. In sum, much nuclear waste is eventually safe (no longer radioactive, radiotoxic, or heavy metal toxic), and some becomes substantially safer but still dangerous (no longer radioactive or radiotoxic but still heavy metal toxic).
By contrast, mining waste never becomes safe. Mining waste contains many of the same radioactive atoms as radioactive waste, including uranium and thorium. These will undergo the same natural decay process and eventually cease to be radioactive, but nearly all decay to toxic heavy metals.
Crucially, radioactive atoms represent a tiny portion of mining waste. Substantially more of the waste is heavy metal toxic (but not radioactive), including lead, cadmium, chromium, copper, nickel, and many others. Because these are not radioactive, they will remain heavy metal toxins forever.
In other words, while nuclear waste becomes less and less dangerous, mining waste does not become safer as time goes on. The heavy metals of mining waste are toxic forever.
#2 Dropping acid
Mining requires a great deal of acid, and there is no equivalent for nuclear waste. For example, producing one ton of lithium requires ten tons of sulfuric acid. This is important for two reasons. First, the acid itself can cause severe chemical burns and is toxic to fish and other water-dwelling plants and animals. Second, heavy metals become substantially more dangerous in acid.
Put another way: uranium and thorium from nuclear waste eventually turn into lead. While no one wants a brick of lead, far more dangerous is that same lead dissolved in acid.
#3 Getting a little wasted vs getting really wasted
By far the biggest difference between nuclear waste and green energy mining waste is the amount of waste produced.
As mentioned above, producing 1 gigawatt of electricity via wind power generates 28,000 tons of radioactive mining waste in the form of radioactive thorium, plus 400,000 cubic meters of heavy metal-bearing waste. Producing a gigawatt of electricity via nuclear generates only 276 tons of radioactive, heavy metal-bearing waste. To give some perspective on the differences in scale, a single modern mine generates hundreds of millions of tons of waste, and phasing out fossil fuels with green energy would require the opening of thousands of new mines. Safely containing hundreds of millions of tons of waste is a massive engineering challenge. Meanwhile, 92 nuclear power plants (54 active and 38 decommissioned) have operated in the US since the 1950s. All nuclear power plants ever operated in the US have generated 90,000 metric tons of waste, or less than one-tenth of 1% of the waste generated by a single modern mine.
Putting this all together, mining waste is substantially more dangerous than spent nuclear fuel. If nuclear power is unacceptable to certain environmentalists due to the issue of radioactive waste, then green energy should also be unacceptable to them. The mining required to make green energy generates substantially more waste overall; waste that is substantially more dangerous; and waste that is substantially more radioactive than that of nuclear power.
A note on timescales
As discussed above, 3% of nuclear waste requires 10,000 years before it ceases to be radiotoxic. And the heavy metals generated by mining never become safe. What would it take to safely store something for 10,000 years? 100,000 years? 1 million years? The timescales involved are literally impossible to imagine, but a thought experiment* can help.
Historians use a cutoff of 500 or 750CE, or about 1500 years ago, to mark the end of ancient history. Let’s use 1500 years as the cutoff for ancient history. In this table, we go back in time in 1500-year increments until we get to 10,000 years, or the amount of time spent nuclear fuel must be safely stored:
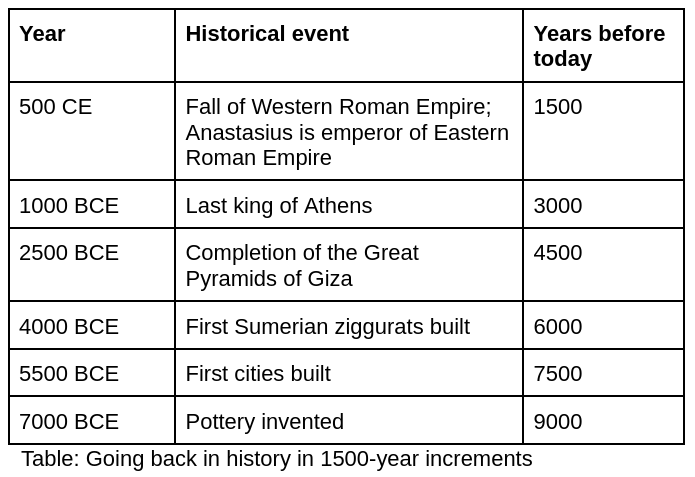
If Emperor Anastasius is ancient history to us because he lived 1500 years ago, then the 1500-year gap means that the last king of Athens is ancient history to Anastasius. And because the Great Pyramids were completed 1500 years earlier, the Great Pyramids were ancient history to the last king of Athens. We have to repeat this 1500-year ancient history exercise six times to get to just 9,000 years back. 10,000 years into the future is the point at which the final 3% of nuclear waste will cease to be radiotoxic.
Clearly, 10,000 years is an extraordinary amount of time. 10,000 years ago was before all recorded history: humanity was still 1,000 years away from inventing pottery and 2500 years away from founding cities. It is beyond hubristic to assume that we can build something that can last for 10,000 years – let alone something so perfectly solid and durable that it will never even develop a crack that a single radioactive atom can seep out of. But for heavy metal toxins, 10,000 years isn’t nearly enough time. 100,000 years isn’t enough time. 1 million years isn’t enough time. Heavy metal toxins never become safe.
The green energy deal with the devil
It gets even worse. The minerals we need in massive amounts to phase out fossil fuels – like lithium, cobalt, nickel, copper, and rare earths – are mostly near the earth’s surface. That means that miners must remove all vegetation and overturn all soil in their search for ore. No ecosystem can survive open-pit mining. Literally thousands of ecosystems will need to be destroyed to obtain these minerals.
And so climate change has forced us to accept a deal with the devil. To save the planet from climate change, it is necessary to destroy parts of the planet. Parts of the planet with minerals needed for a green energy transition will have to be sacrificed by way of mining waste and open-pit mining for the rest of the planet to survive. There is no other way, we are told, to replace fossil fuels.
But what if this Faustian bargain is a lie?
Sign up here to be notified when a new issue of Finite is published (4 emails/year)
As we’ll see in this issue, there are three major ways that this deal with the devil is a lie. First, mining is a massive contributor to greenhouse gas emissions, accounting for an estimated 10% of humanity’s greenhouse gas emissions. These emissions have two sources. First, mining and processing ore require fossil fuels, and most of these activities cannot be decarbonized. Second, the deforestation of open-pit mining generates enormous greenhouse gas emissions. When all the vegetation and microscopic organisms are burned or die off and decay, a tremendous amount of greenhouse gas is released. This is analogous to our discussion of land degradation in Issue 1. As we saw in Issue 1, a quarter of humanity’s greenhouse gas emissions result from land degradation. Once in the atmosphere, the greenhouse gases from desertification or deforestation are no different from greenhouse gases from any other source. Whether released by a fossil fuel power plant or decaying trees, greenhouse gases have the same dangerous, planet-warming effect. Only some fossil fuel use in mining can be eliminated; deforestation cannot be eliminated. In other words, while scientists are unanimous that we must fully eliminate greenhouse gas emissions, we actually cannot do so if we must continually mine to build, repair, and replace green energy infrastructure into the future.
The second way that this Faustian bargain is a lie is that we’re not sacrificing parts of the planet that happen to have deposits of critical minerals; we’re sacrificing parts of the planet that happen to have poor and powerless people who can’t stand up to mining corporations. As we’ll see in Chapter 2, the highest quality nickel ore is found in the US, Canada, and Russia, but the world’s biggest producer of nickel is Indonesia. As a technical matter, it makes no sense to use Indonesian nickel because it requires far more energy to process into a usable form. But, as a political matter, it makes perfect sense: no one wants a mine opened near their home, so mines are more likely to be opened where people lack the power to fight back. Similarly, rare earths are relatively uniformly distributed around the earth’s surface, but mining is concentrated in Myanmar, where impoverished locals and a weak, corrupt government are unable – or unwilling – to prevent illegal mining.
The third reason this Faustian bargain is a lie is that we don’t need all the energy we think we do. Our economies are so grossly inefficient that we could reduce our energy use while improving our quality of life. As we’ll see in Chapters 3 and 4, there is no better example than electric cars.
By the end of Chapter 4, a way forward will be clear: how we can phase out fossil fuels while minimizing mining and improving our quality of life.
*Inspired by an engrossing article by Aisling McCrea